 When a government thinks they can control the supply of goods better than the Free Market then supply shortages and price controls are sure to follow. We have seen throughout history when countries try to tinker with the Free Market and implement varying degrees of Socialism that bureaucrats fail at this attempt. The Invisible Hand, first characterized by Adam Smith, states that the profit motive of individuals will seek to provide a good or service to the Market in the most efficient manner and doesn’t need a team of czars directing their actions. The Free Market economic system has propelled the United States to a world leader and we didn’t need hundreds of czars to oversee the means of production for all industries.
When a government thinks they can control the supply of goods better than the Free Market then supply shortages and price controls are sure to follow. We have seen throughout history when countries try to tinker with the Free Market and implement varying degrees of Socialism that bureaucrats fail at this attempt. The Invisible Hand, first characterized by Adam Smith, states that the profit motive of individuals will seek to provide a good or service to the Market in the most efficient manner and doesn’t need a team of czars directing their actions. The Free Market economic system has propelled the United States to a world leader and we didn’t need hundreds of czars to oversee the means of production for all industries.
Supply shortages and price controls can’t happen in American though, right? Well, it is already starting and with goods that are necessary to treat our sick children.
There was a story on NPR today showing how critical children’s Leukemia drugs are in scarce supply.
“The latest crisis concerns the old standby cancer drug methotrexate. For six decades, it has made the difference between rapid death and lifelong cure for thousands of children with acute lymphoblastic leukemia, or ALL, and a type of bone cancer called osteogenic sarcoma.”
“Many hospitals around the nation are perilously close to running out of a form of methotrexate that is necessary to inject in high doses to treat these forms of cancer.”
“For ALL and osteogenic sarcoma, “there’s really no known curative therapy without methotrexate,” says Dr. Howard Weinstein of Massachusetts General Hospital. “Based on our current outlook, if we don’t get any supply, we’re going to be out of methotrexate in the next couple of weeks.”
“We’re probably good for a few weeks, but if we don’t get any, we’ll have to do some substitution or delays of therapy and give the high-dose methotrexate later when it’s available,” says Dr. Bruce Bostrom.”
“He says doctors just don’t know how that’s going to affect patients’ chance of a cure — which is at least 90 percent with the right treatment.”
“Brenda Carr’s 4-year-old daughter, Rowen, is a patient at Bostrom’s hospital.”
“What I just couldn’t believe was that there was nothing really to prevent this from happening,” Carr says. “It gets really frustrating. I try to stay logical about it, but, unfortunately, I’m a mom.”
How did we get to this point where a high demand drug is in scarce supply? From the NPR story:
“The reason for this particular shortage is that a principal supplier of injectable methotrexate, Ben Venue Laboratories of Bedford, Ohio, shut down in November after it flunked an inspection by the Food and Drug Administration.”
“Erin Fox, an expert on drug shortages at the University of Utah, says the FDA inspectors found a long list of serious problems.”
“If you want to read something to give you nightmares, you can look at the FDA 483 inspection form,” Fox says. “You can read about mold on the walls and rust from machinery falling into the vials. It really provides a very grim picture of a crumbling factory.”
“The same lab is responsible for another recent and far-reaching drug shortage involving Doxil, a drug relied on by thousands of patients with ovarian and breast cancer. Ben Venue Laboratories was the sole worldwide supplier of Doxil, which is now unobtainable.”
Well, I don’t blame the FDA for shutting down this facility if these conditions truly did exist but for a drug that is such high demand, surely there is another supplier who is in the Market to take advantage of this other company’s failure.
“Valerie Jensen of the FDA’s office of drug shortages says three other manufacturers have been persuaded to ramp up production of preservative-free methotrexate.”
That is good news but I’m curious why the FDA needed to persuade them to ramp up production. We’ll get to that later but what about the other critical drug, Doxil, from that troubled lab?
“Jensen said the FDA expects to have good news about Doxil. She says the agency is exploring the possibility of licensing a foreign company to make the drug, something the FDA has done eight times in the past year with other drugs in critical shortage.”
What? We have to use a foreign company to manufacture this drug? Why aren’t there any US companies that are already producing this drug? We have only one company in the whole US that can produce this drug?
It gets worse.
“Carr, the Minneapolis mom, says she has her fingers crossed. But she’s skeptical.”
“This isn’t the first time we’ve been on the brink of drug shortages, especially in cancer treatments,” Carr says. “I don’t want another mom to have to be thinking about whether or not their child is going to get the medication they need.”
“Experts say there certainly will be other parents who will have to worry about other shortages, because the root causes will not be easily resolved.”
Why do we, all of a sudden, see massive drug shortages in the US? There is obviously a market for these drugs so why are companies not producing them? What are these ‘root causes’ that the NPR article speaks of? Since the article didn’t elaborate, let me take a swipe at it.
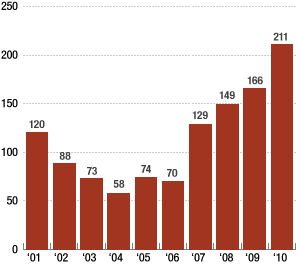
From another NPR article, the number of drug shortages per year is plotted on the following graph.
Since 2007 we have seen an increase in the number of drug shortages. Why would such a lucrative market see an exit of companies willing to compete? Could the over regulation of the FDA have something to do with it?
This paper from the Progressive Policy Institute (PPI), which was written in June 2011, seems to think so.
“For the purposes of this paper, however, we will focus on the FDA, which has been criticized for imposing excessive requirements on the approval of new drugs and medical devices. Three facts are clear. First, the FDA’s regulatory reach and intensity has increased over the past 10 years. FDA employment grew by 33 percent between 2000 and 2011, even as employment in the regulated industries – pharmaceuticals, medical devices, and biotech – only rose by 3 percent.”
“Second, in the wake of high-profile episodes such as the Vioxx case, the FDA has gotten stricter about requiring evidence of safety and effectiveness before approving new drugs. Third, the number of new drugs approved fell sharply over the past decade compared to the decade before.”
I wrote a post last year showing how over regulation from the US government is literally killing us and this article by the PPI validates my claims. It basically comes down to this – Why would a company waste resources (and several millions of dollars) to develop and validate a drug only to experience road blocks from the FDA that hinder the product coming to market? The answer is, they won’t.
This is what we get from government over stepping its bounds with regard to regulation of the market and this is just the beginning unless the elections of 2012 bring about real change that we can believe in.


You are confusing criticism of the FDA for “imposing excessive requirements on the approval of new drugs” with the inspection of plants producing drugs like methotrexate that have been approved and on the market for years.
As a physician, I’m very glad we have the FDA to do these inspections. The private companies that make the medicines are apparently in at least some cases doing a terrible job of quality control. The inspection report on Ben Venue’s plant which shut down because of quality problems is a public record: http://www.fda.gov/downloads/AboutFDA/CentersOffices/OfficeofGlobalRegulatoryOperationsandPolicy/ORA/ORAElectronicReadingRoom/UCM282550.pdf
Read the report and see if you don’t agree we should be thanking the FDA for doing its job.
As far as the magic of the market goes. That is historically and empirically an iffy thing. Sometimes there are serious market failures. In this case it should be noted that almost all of the medicine shortages in the US in recent years involve generic medicines where the free market sets the price. The problem is pharmaceutical manufacturers are used to huge (some would say excessive) profits on patented drugs, ie. where a monopoly environment determines the price. The buyers in this case are patients, many of whom depend on the various medicines for their lives. They are not in a strong position to push back against excessive prices.
The available remedy for market failures is — like it or not — the government of the people.
We need to support and celebrate it when it is working, as it clearly did here.
Doug McVicar, MD
Wonalancet, NH
Dr. McVicar, Thanks for stopping by and posting your thoughts. Much appreciated.
I agree with you in the closing of the Ben Venue’s plant and even stated so in my post above. This is what the FDA needs to be doing and I echo your praise of that.
For the rest of your post…. we’ll have to agree to disagree. Did you check out the PPI link in the article? I can echo the findings in this paper based on what I’ve seen over the past 3-4 years with regard to regulatory scrutiny of new products. It is real, it is happening and it is affecting the supply to the market. Drugs rolling off brand name and into generic is not something that just started in 2007 but drug shortages starting occurring more in this year and have increased every year since.
If you have a link to share showing that this increase in drug shortages is correlated to increased generics I’d like to see that and this data might change my opinion. It won’t change my opinion of the over zealous FDA regulation I see in my industry because I see that with my own eyes but it might change my opinion of the pharma arena.
Thanks again for stopping by!
Pingback: The Reality of Socialism | cosmoscon